Institute of Biomedical Research and InnovationDepartment of Regenerative Medicine Research
Research Overview
Establishment of optimized stem cell therapy for stroke and dementia through enhancing endogenous neurogenesis
Stroke and dementia are the leading and second cause of bedridden patients, respectively. Currently, there are no effective treatments to cure these diseases. We have shown that (1) intra-venous injection of stem cells for stroke improves functional recovery through activation of neurogenesis at peri-infarct area, (2) injection of stem cells for dementia improves neuronal function through activation of neurogenesis at hippocampus, and (3) the fundamental mechanism of action of stem cell therapies, including hematopoietic and mesenchymal stem cell therapies, is direct cell-cell interaction via gap junctions (Fig. 1). Based on these findings, we are currently conducting research to establish optimized stem cell therapy for stroke and dementia.
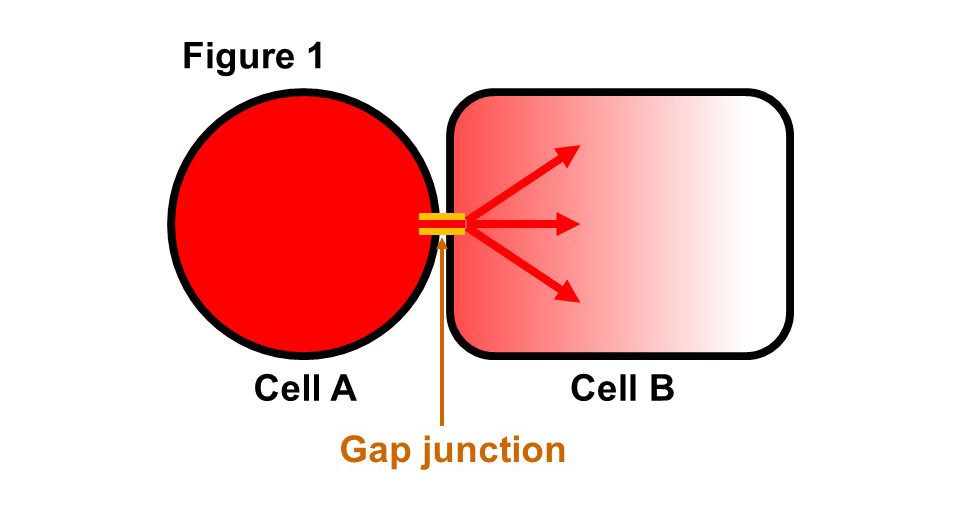
(Description of Fig. 1)
- Fig. 1 Schematic diagram of gap junction Gap junction is an intercellular tunnel that connect cytosols of contacting cells. Small molecules and ions with molecular weights of less than 1500 can pass freely into the cytoplasm of neighboring cells according to a concentration gradient thought gap junction.
NEWS
Member
- Professor
- Akihiko Taguchi,
M.D., Ph.D.
- @akitaguchi_lab(External link)
- taguchi★fbri.org
(Replace ★ with @)

- Research focus
- Publications
Research focus
- (1) Establishment of optimized stem cell therapy for stroke
Our Achievements
We have established a highly reproducible stroke mouse model which is ideal for the evaluation of various cell therapies. Using this model, we have found that hematopoietic stem cell transplantation after stroke enhances cerebral angiogenesis and the angiogenesis is essential for endogenous neurogenesis after stroke (Taguchi et al. J. Clin. Invest. 2004) (Fig. 2).
(b) Stem cell transplantation improves neurological recovery in patients with stroke
We conducted a clinical trial of autologous hematopoietic stem cell transplantation for patients with severe stroke. We have found that stem cell transplantation improves functional recovery with increased cerebral metabolism (Taguchi et al. Stem Cells Dev. 2015) (Fig. 3).
(c) Identification of the mechanism of action of stem cell therapy
We have revealed that the mechanism of action of hematopoietic stem cells is direct cell-cell interaction via gap junctions (Fig. 4) (Kikuchi-Taura et al. Stroke 2020). Furthermore, we have revealed that mechanism of action of mesenchymal stem cells is also direct cell-cell interaction via gap junctions (Fig. 5) (Kikuchi-Taura et al. StemCells 2021).
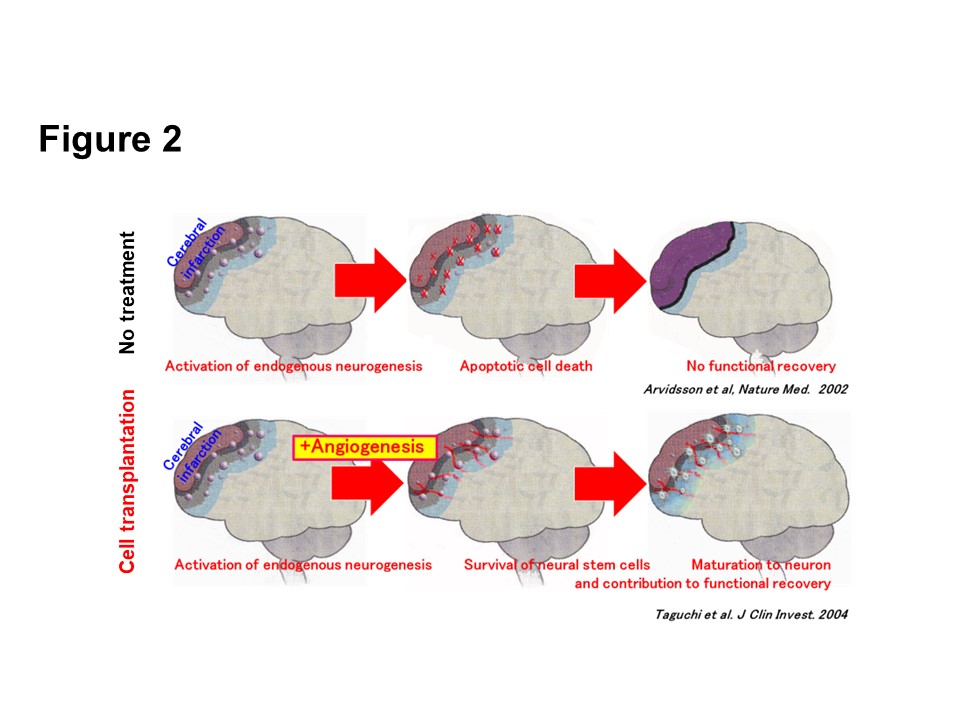
(Description of Fig. 2)
- Fig. 2 Therapeutic angiogenesis for endogenous neurogenesis
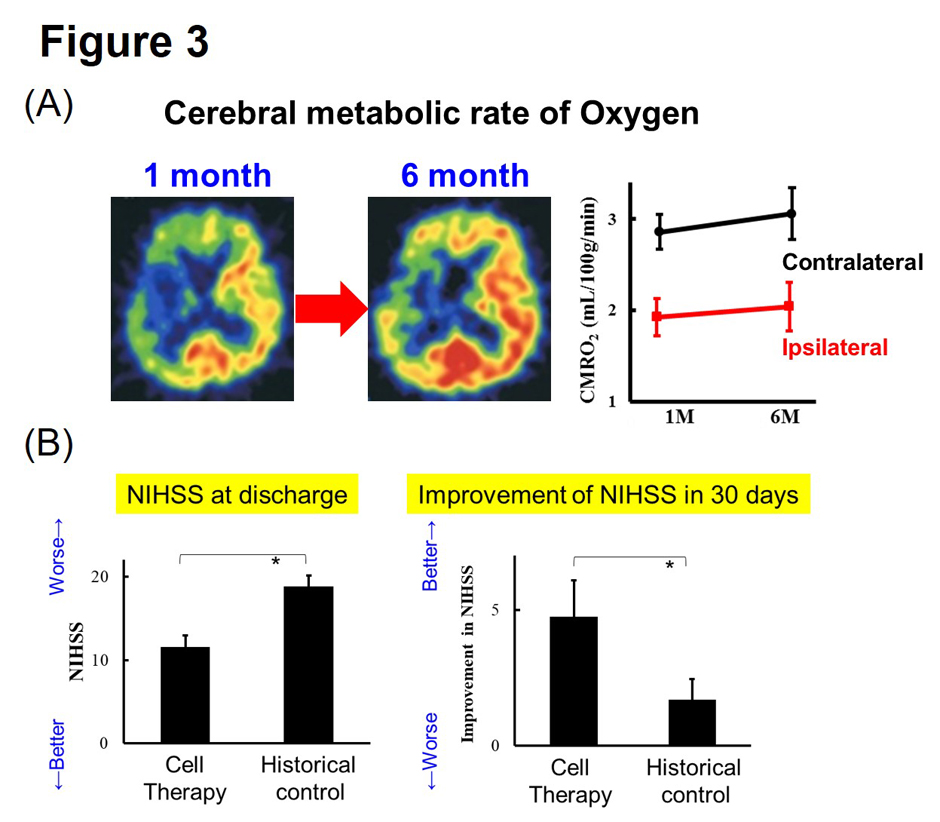
(Description of Fig. 3)
- Fig. 3 Improvement of cerebral metabolism and neurological function by autologous hematopoietic stem cell transplantation in stroke patients
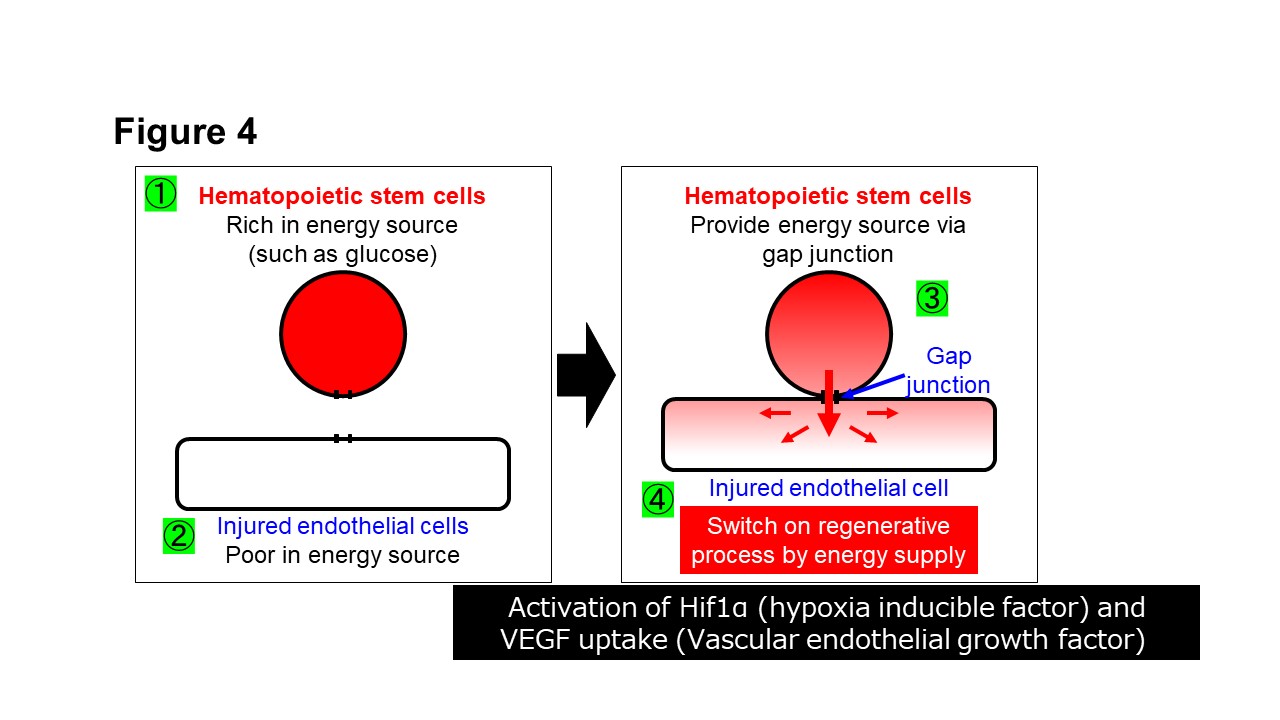
(Description of Fig. 4)
- Fig. 4 Mechanism of action of hematopoietic stem cell therapy
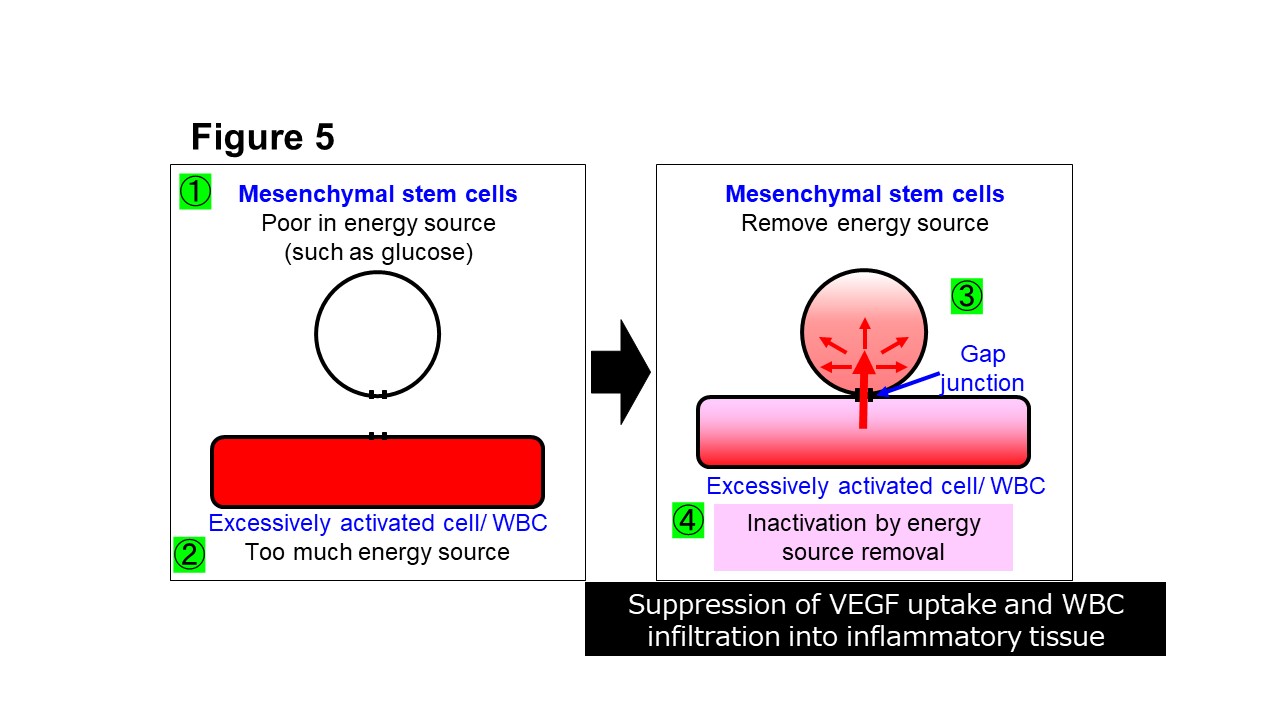
(Description of Fig. 5)
- Fig. 5 Mechanism of action of mesenchymal stem cell therapy
- Current projects
Stem cell therapy is not yet widely used as a general medical treatment due to its uncertain efficacy and extremely high cost. Our discoveries about the fundamental mechanism of action of stem cell therapies enable the development of stem cell products with reliable therapeutic effects that can be widely used in medical economics. Currently, we are conducting research for optimization of stem cell therapy, including the full elucidation of molecules that move through gap junctions, identification of gap junction inhibitor, search for substances that improve stem cell function, and establishment of quantitative evaluation methods of stem cell function.
(b) Research for small molecules and liposomes that substitute stem cell therapy
The findings about mechanism of action of stem cell therapy enable to develop small molecules and liposomes that can substitute stem cell therapy. Currently, projects of constructing functional liposomes and searching for low molecular weight compounds that can substitute for the action of stem cell therapy are ongoing.
- (2) Establishment of optimized stem cell therapy for dementia
Our Achievements
Simple aging mouse is an ideal model that accurately reflects the pathology of human dementia patients in elderly, as they show decreased motor skills, muscle mass, immunocompetence, wound-healing ability and cognitive decline. We have found that transplantation of hematopoietic stem cells to aged mice improves short term memory (Takeuchi et al. Front Aging Neurosci. 2020) with enhanced neurogenesis at hippocampus (Fig. 6) (Takeuchi et al. Front Aging Neurosci. 2022)
(b) Age-related decline in function of gap junction and its activation by stem cells.
We have found that the expression of gap junction is decreased with aging in hippocampus and stem cells transplantation increases the expression of gap junction (Takeuchi Front Aging Neurosci. 2022).
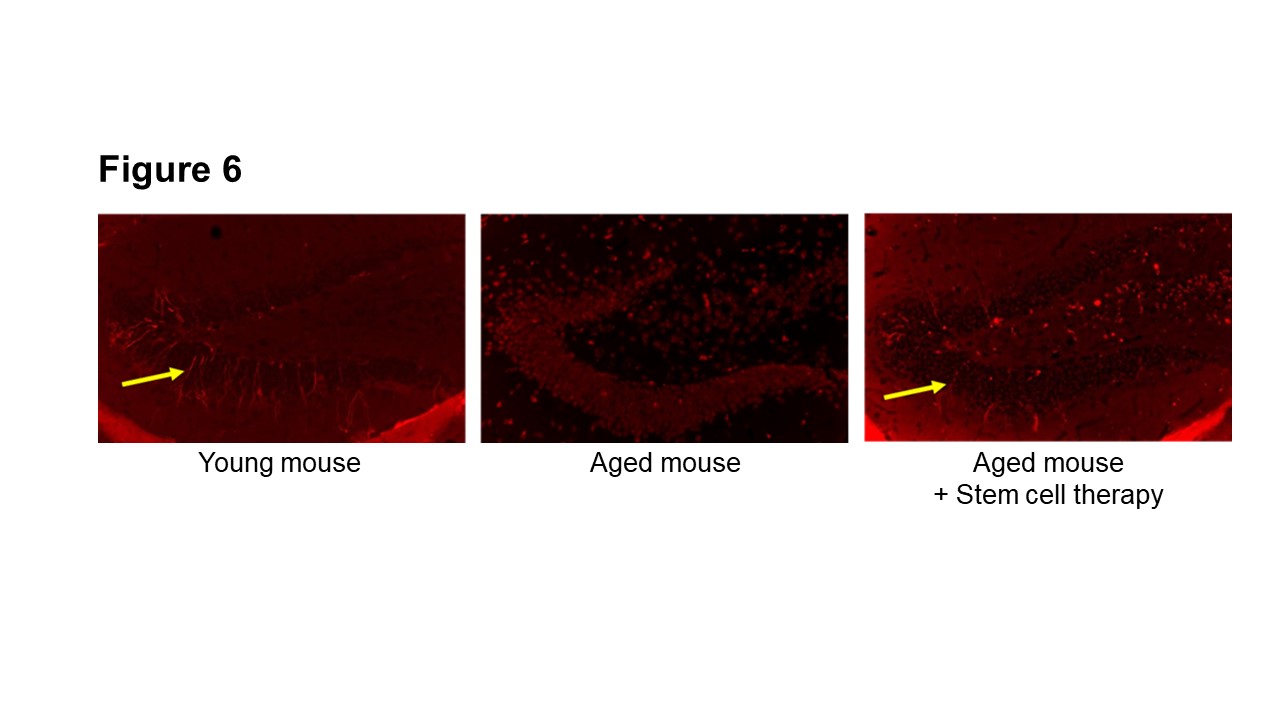
(Description of Fig. 6)
- Fig. 6 Activation of neurogenesis at hippocampus by hematopoietic stem cell transplantation in aged mice
- Current projects
(b) Elucidation of the nature of aging
Cell-cell interaction via gap junctions has significant role in multicellular organisms. Our findings indicated that cell-cell interaction via gap junctions is significantly impaired with aging and it is activated by stem cells transplantation. These findings indicate that cell-cell interactions via gap junctions would have a significant impact on aging in multicellular organisms. We are conducting research to elucidate the nature of aging from the viewpoint of gap junctions.

(Description of Fig. 7)
- Fig. 7 Our therapeutic strategy for Alzheimer’s disease
Achievements
- Nabetani M, Mukai T, Taguchi A. Cell therapies for autism spectrum disorder based on new pathophysiology: A review. Cell Transplant. 2023 Jan-Dec;32:9636897231163217.
- Nakano-Doi A, Kubo S, Sonoda E, Taguchi A, Nakagomi T. Different contacted cell types contribute to acquiring different properties in brain microglial cells upon intercellular interaction. Int J Mol Sci.2023 Jan 16;24(2):1774. doi: 10.3390/ijms24021774.
- Ogawa Y, Akamatsu R, Fuchizaki A, Yasui K, Saino O, Tanaka M, Kikuchi-Taura A, Kimura T, Taguchi A. Gap junction-mediated transport of metabolites between stem cells and vascular endothelial cells. Cell Transplant. 2022 Jan-Dec;31:9636897221136151. doi: 10.1177/09636897221136151.
- Ogawa Y, Okinaka Y, Kikuchi-Taura A, Saino O, Tani-Yokoyama A, Masuda S, Komatsu-Horii M, Ikemoto Y, Kawamoto A, Fukushima M, Taguchi A. Pre-clinical proof of concept: Intra-carotid injection of autologous CD34 positive cells for chronic ischemic stroke, Front Med, 2022. Mar 11;9:681316. doi: 10.3389/fmed.2022.681316.
- Takeuchi Y, Saino O, Okinaka Y, Ogawa Y, Akamatsu R, Kikuchi-Taura A, Kataoka Y, Maeda M, Gul S, Claussen C, Boltze J, Taguchi A. Increased RNA transcription of energy source transporters in circulating white blood cells of aged mice. Front Aging 2022. Feb 3;14:759159. doi: 10.3389/fnagi.2022.759159.
- Okamoto T, Park EJ, Kawamoto E, Usuda H, Wada K, Taguchi A, Shimaoka M. Endothelial connexin-integrin crosstalk in vascular inflammation. Biochim Biophys Acta Mol Basis Dis. 2021;1867(9):166168.
- Ogawa Y, Saino O, Okinaka Y, Takeuchi Y, Saino O, Kikuchi-Taura A, Taguchi A. Bone Marrow Mononuclear Cells Transplantation and Training increased transplantation of energy source transporters in chronic stroke, JSCVD, 2021. Jun 17;30(8):105932. doi: 10.1016/j.jstrokecerebrovasdis.2021.105932.
- Kikuchi-Taura A, Okinaka Y, Saino O, Takeuchi Y, Ogawa Y, Kimura T, Gul S, Claussen C, Boltze J, Taguchi A. Gap junction-mediated cell-cell interaction between transplanted mesenchymal stem cells and vascular endothelium in stroke. Stem Cells. 2021 Mar 11. doi: 10.1002/stem.3360.
- Boltze J, Taguchi A, 他. New Mechanistic Insights, Novel Treatment Paradigms, and Clinical Progress in Cerebrovascular Diseases. Front Aging Neurosci. 2021 Jan 28;13:623751. doi: 10.3389/fnagi.2021.623751.
- Ogawa Y, Okinaka Y, Takeuchi Y, Saino O, Kikuchi-Taura A, Taguchi A. Intravenous Bone Marrow Mononuclear Cells Transplantation Improves the Effect of Training in Chronic Stroke Mice. Front. Med., November 26, 2020. doi: 10.3389/fmed.2020.535902.
- Boltze J, Didwischus N, Li S, Jolkkonen J, Taguchi A. Increased migratory and Homing abilities of neural and mesenchymal stem cell populations by transient cell modifications: Preclinical progress and clinical relevance. EBioMedicine. 2020 Sep 24;60:103022. doi: 10.1016/j.ebiom.2020.103022.
- Takeuchi Y, Okinaka Y, Ogawa Y, Kikuchi-Taura A, Kataoka Y, Gul S, Claussen C, Boltze J, Taguchi A. Intravenous Bone Marrow Mononuclear Cells Transplantation in Aged Mice Increases Transcription of Glucose Transporter 1 and Na+/K+-ATPase at Hippocampus Followed by Restored Neurological Functions. Front. Aging, June 11, 2020. doi: 10.3389/fnagi.2020.00170.
- Kikuchi-Taura A, Okinaka Y, Takeuchi Y, Ogawa Y, Maeda M, Kataoka Y, Yasui T, Kimura T, Gul S, Claussen C, Boltze J, Taguchi A. Bone marrow mononuclear cells activate angiogenesis via gap junction mediated cell-cell interaction. Stroke. 2020.51(4):1279-1289. doi: 10.1161/STROKEAHA.119.028072.
Japanese reviews
- Yuko Ogawa, Akihiko Taguchi, “Advances in Stroke Regenerative Medicine,” Igaku no Ayumi, 280 (10), 1099-1103 (2022).
- Akihiko Taguchi, Yuko Ogawa, “Cerebral Vascular Endothelial Cells in Stem Cell Therapy,” the latest Clinical Stroke Studies (2nd edition), 80, 639-643 (2022)
- Akihiko Taguchi,Regenerative medicine for stroke. Taguchi A. The Japanese Journal of Physical Therapy. 2017;51(5):451-456
- Akihiko Taguchi,Cell based therapy for stroke patients. Taniguchi A. Journal of Clinical and Experimental Medicine. 2017;260(7):563-567
- Akihiko Taguchi, Regenerative Medicine for Stroke. PT Journal. 2017; 51(5):451-456
- Akihiko Taguchi, Regenerative medicine for stroke. Taguchi A. Medicina. 2016;53(2):340-342
- Akihiko Taguchi, Stroke and biomarkers: New development; Hematopoietic stem cell count in peripheral blood. Taguchi A. Molecular Cerebrovascular Medicine. 2016;15(2):17-21
- Akihiko Taguchi, Autologous bone marrow monocyte transplantation for cerebral infarction. Taguchi A. Clinical Neuroscience. 2016;34(11):1261-1262
Major Awards in Recent Years
- Yuko Ogawa “Development of Allogeneic Hematopoietic Stem Cell Products for Regenerative Medicine,” Excellent Presentation Award, the 64th Annual Meeting of the Japanese Society of Cerebral Blood Flow and Metabolism


