It is needless to say that cell and gene therapy will be indispensable medical technology in a near future. For its industrialization and to maximize the productivity, it is necessary to form an ecosystem with not only CMO/CDMO but also cell manufacturing industry at its core. The Research & Development Center for Cell Therapy (RDC) at FBRI suggests the cell manufacturing ecosystem.
Having established a PIC/S GMP-compliant manufacturing facility for gene and cell preparations in Kobe, we are well aware of the conditions and issues necessary for the continuous establishment of cell manufacturing industry. Since an industrial ecosystem is already in place in Kobe, we are ready to welcome new CMO/CDMO pipelines.
If we can efficiently manufacture cell and gene products, not only at the commercial manufacturing stage but also at the start-up of facilities, we can expect a new growth industry. Kobe can be a role model for such new growth industry around the world.
Take a look at this introduction video and find out about Cell Port City, a cutting-edge cell and gene therapy and cell manufacturing ecosystem in KBIC.
URL https://www.youtube.com/watch?v=0iuWBPQpuMQ

*Integrated system from basic research to R&D, and to commercialization

*Cutting-edge cell processing facility
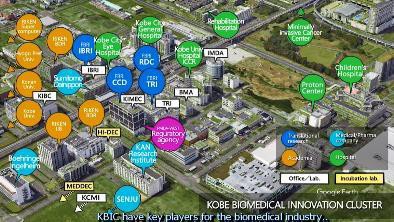
*KBIC have key players for the biomedical industry

*Automated cell culture system with monitoring

*Formation of Cell Port City for sustainable growth