The Foundation for Biomedical Research and Innovation at Kobe (FBRI, President: Nobel laureate Dr. Tasuku Honjo) announced the discovery by their researchers of the Department of Immunology (Prof. Akio Ohta) for the distinct class of anti-PD-1 antibodies in collaboration with Meiji Seika Pharma Co., Ltd., National Institutes of Biomedical Innovation, Health and Nutrition (NIBIOHN), RIKEN and Kyoto University. Their findings were published in Science Immunology on January 13, 2023.
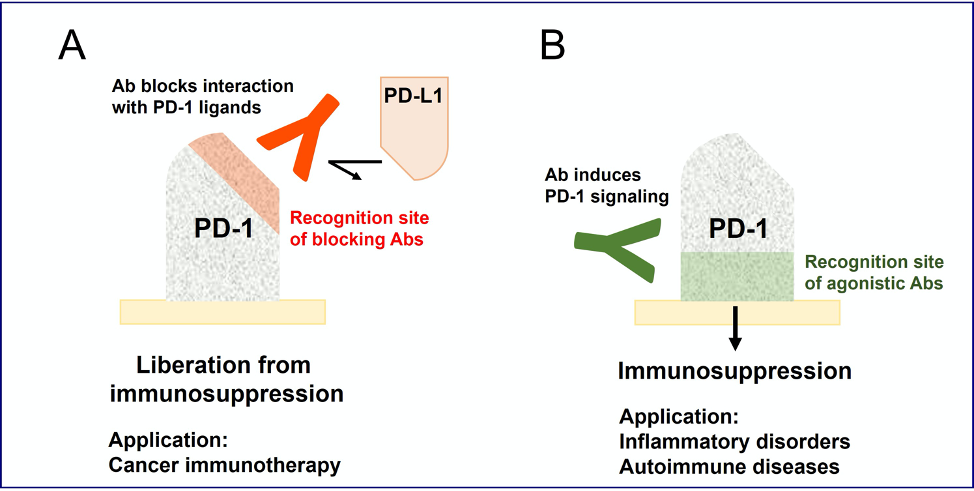
Anti-PD-1 antibodies (A) that are in practical use for cancer immunotherapy block immunosuppressive signaling and enhance immune response. In sharp contrast, the new class of anti-PD-1 antibodies (B) in the current study recognize a different region from blocking antibodies and can trigger immunosuppressive signaling.
Summary
- ・PD-1 represents physiological mechanisms of negative immune regulation. The Nobel Prize was awarded in 2018 for the discovery of cancer therapy by blocking negative immune regulators.
- ・While anti-PD-1 antibodies for cancer treatment are immunoenhancing, the current study revealed a distinct group of antibodies with an opposite effect, turning on the immunosuppressive activity of PD-1.
- ・These PD-1 agonist antibodies may open a path for the novel treatment of inflammatory disorders including autoimmune diseases.
1 Research Background
PD-1 is expressed on activated lymphocytes and negatively modulates the intensity of immune response. The biological significance of PD-1-dependent immunosuppression in the human immune system has been demonstrated by the application of anti-PD-1 antibody to the successful cancer treatment. The blockade of PD-1-dependent immunosuppression leads to enhanced anti-tumor immunity, thereby enforces tumor regression. Such immunoenhancing anti-PD-1 blocking antibodies have been known for 20 years; however, agonistic antibodies that can stimulate the immunosuppressive activity of PD-1 are yet to be defined. Pharmacological PD-1 stimulation by agonist antibodies is potentially an effective treatment to downregulate overwhelming inflammatory activities in various diseases. Several pharmacological companies are engaged in the development of immunosuppressive anti-PD-1 agonist antibodies.
2 Research Significance and Future Development
In 2016, the Department of Immunology of FBRI started the research on anti-PD-1 agonist antibody in a collaboration with Meiji Seika Pharma Co., Ltd. We started from a question how it would become possible to stimulate PD-1 using immunoglobulins. To our finding, one important requirement for agonist antibodies was binding to the membrane-proximal region of PD-1 molecule. Another requirement to trigger PD-1 signaling was the immobilization of agonist antibodies on the antigen-presenting cells. Fc receptors mediate the immobilization of anti-PD-1 agonist antibodies and enable PD-1 stimulation.
Based on these findings, we established the screening system for the agonistic activity of anti-human PD-1 antibodies. Through a screening campaign with groups of antibodies made by the patented epitope-normalized antibody panel (ENAP) technlogy at NIBIOHN, we found quite a few agonist antibodies from the screening of 81 monoclonal antibody clones. All the agonist antibodies bound to the membrane-proximal region, which is in sharp contrast to the recognition of the membrane-distal region by blocking antibodies. Since the group of agonist antibodies is clearly distinct from blocking antibodies, anti-PD-1 agonist antibodies may have not been recognized for a long time behind the strong emphasis on the development of blocking antibodies for cancer immunotherapy.
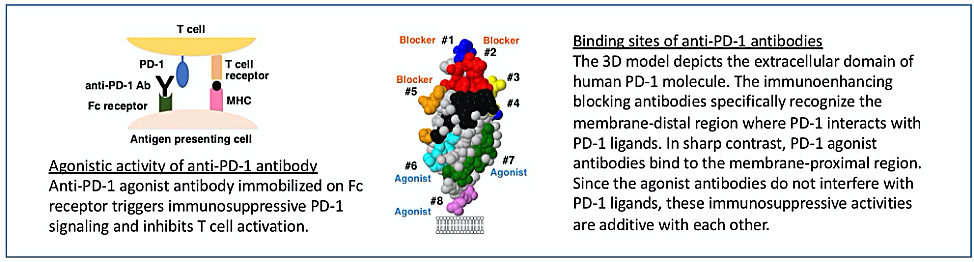
We confirmed the anti-inflammatory efficacy of the PD-1 agonist antibody in various animal models of T cell-dependent inflammatory disease. Furthermore, we found that the increased Fc receptor binding of PD-1 agonist antibody contributes to enhanced immunosuppressive activity. Indeed, protein engineering on the agonist antibody substantially improved its immunosuppressive potential against human T cells.
PD-1-dependent immunoregulation is involved in different types of immune responses including T cell-dependent tissue injury and autoantibody production. Therefore, anti-PD-1 agonist antibodies may possibly be developed into a versatile therapeutic option to a wide range of inflammatory disorders. PD-1 has been a target molecule for the development of immunosuppressants. The current study defined specific requirements for anti-PD-1 agonist antibodies. These findings may contribute to the mechanism-based development of therapeutics.
In collaboration with Meiji Seika Pharma, we will continue the development of anti-human PD-1 agonist antibody for clinical application. We have applied for patents regarding anti-human PD-1 agonist antibodies.
3 Article Information
Title: Anti-PD-1 antibodies recognizing the membrane-proximal region are PD-1 agonists that can downregulate inflammatory diseases
Authors: Kensuke Suzuki1,2, Masaki Tajima1, Yosuke Tokumaru1,2, Yuya Oshiro1,2, Satoshi Nagata3, Haruhiko Kamada3, Miho Kihara4, Kohei Nakano4, Tasuku Honjo5 and Akio Ohta1
Affiliations:
- 1 Department of Immunology, Institute of Biomedical Research and Innovation, Foundation for Biomedical Research and Innovation at Kobe, Kobe 650-0047, Japan
- 2 Pharmaceutical R&D Division, Meiji Seika Pharma Co., Ltd., Tokyo 104-8002, Japan
- 3 Laboratory of Antibody Design, Center for Drug Design Research, National Institutes of Biomedical Innovation, Health and Nutrition, Ibaraki 567-0085, Japan
- 4 Laboratory for Animal Resources and Genetic Engineering, RIKEN Center for Biosystems Dynamics Research, Kobe 650-0047, Japan
- 5 Department of Immunology and Genomic Medicine, Center for Cancer Immunotherapy and Immunobiology, Graduate School of Medicine, Kyoto University, Kyoto 606-8507, Japan
- Journal: Science Immunology
- DOI: 10.1126/sciimmunol.add4947
Corresponding Author
Akio Ohta, Ph.D.

Professor
Department of Immunology,
Institute of Biomedical Research and Innovation,
Foundation for Biomedical Research and Innovation at Kobe
Email: ohta-a (Please add “@fbri.org” to the end)
https://www.fbri-kobe.org/english/laboratory/research1/
Further Information or Media Contact
Public relations and Strategic planning group,
Foundation for Biomedical Research and Innovation at Kobe
Email:kbic-pr (Please add “@fbri.org” to the end)